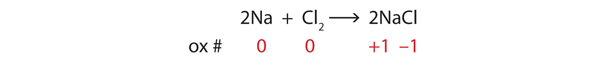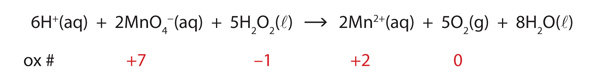5.6 Oxidation-Reduction Reactions
Learning Objectives
- Define oxidation and reduction.
- Assign oxidation numbers to atoms in simple compounds.
- Recognize a reaction as an oxidation-reduction reaction.
Consider this chemical reaction:
2 Na(s) + Cl2(g) → 2 NaCl
The reactants are elements, and it is assumed that they are electrically neutral; they have the same number of electrons as protons. The product, however, is ionic; it is composed of Na+ and Cl− ions. Somehow, the individual sodium atoms as reactants had to lose an electron to make the Na+ ion, while the chlorine atoms as reactants had to each gain an electron to become the Cl− ion. This reaction involves the transfer of electrons between atoms.
In reality, electrons are lost by some atoms and gained by other atoms simultaneously. However, mentally we can separate the two processes. Oxidation is defined as the loss of one or more electrons by an atom. Reduction is defined as the gain of one or more electrons by an atom. In reality, oxidation and reduction always occur together; it is only mentally that we can separate them. Chemical reactions that involve the transfer of electrons are called oxidation-reduction (or redox) reactions.
Redox reactions require that we keep track of the electrons assigned to each atom in a chemical reaction. How do we do that? We use an artificial count called the oxidation number to keep track of electrons in atoms. Oxidation numbers are assigned to atoms based on a series of rules. Oxidation numbers are not necessarily equal to the charge on the atom; we must keep the concepts of charge and oxidation numbers separate.
The rules for assigning oxidation numbers to atoms are as follows:
- Atoms in their elemental state are assigned an oxidation number of 0.
- Atoms in monatomic (i.e., one-atom) ions are assigned an oxidation number equal to their charge. Oxidation numbers are usually written with the sign first, then the magnitude, which differentiates them from charges.
- In compounds, fluorine is assigned a −1 oxidation number; oxygen is usually assigned a −2 oxidation number (except in peroxide compounds [where it is −1] and in binary compounds with fluorine [where it is positive]); and hydrogen is usually assigned a +1 oxidation number (except when it exists as the hydride ion, H−, in which case rule 2 prevails).
- In compounds, all other atoms are assigned an oxidation number so that the sum of the oxidation numbers on all the atoms in the species equals the charge on the species (which is zero if the species is neutral).
Let us work through a few examples for practice. In H2, both hydrogen atoms have an oxidation number of 0, by rule 1. In NaCl, sodium has an oxidation number of +1, while chlorine has an oxidation number of −1, by rule 2. In H2O, the hydrogen atoms each have an oxidation number of +1, while the oxygen has an oxidation number of −2, even though hydrogen and oxygen do not exist as ions in this compound as per rule 3. By contrast, by rule 3 in hydrogen peroxide (H2O2), each hydrogen atom has an oxidation number of +1, while each oxygen atom has an oxidation number of −1. We can use rule 4 to determine oxidation numbers for the atoms in SO2. Each oxygen atom has an oxidation number of −2; for the sum of the oxidation numbers to equal the charge on the species (which is zero), the sulfur atom is assigned an oxidation number of +4. Does this mean that the sulfur atom has a 4+ charge on it? No, it only means that the sulfur atom is assigned a +4 oxidation number by our rules of apportioning electrons among the atoms in a compound.
Example 13
Assign oxidation numbers to the atoms in each substance.
- Br2
- SiO2
- Ba(NO3)2
Solution
- Br2 is the elemental form of bromine. Therefore, by rule 1, each atom has an oxidation number of 0.
- By rule 3, oxygen is normally assigned an oxidation number of −2. For the sum of the oxidation numbers to equal the charge on the species (which is zero), the silicon atom is assigned an oxidation number of +4.
-
The compound barium nitrate can be separated into two parts: the Ba2+ ion and the nitrate ion. Considering these separately, the Ba2+ ion has an oxidation number of +2 by rule 2. Now consider the NO3− ion. Oxygen is assigned an oxidation number of −2, and there are three oxygens. According to rule 4, the sum of the oxidation number on all atoms must equal the charge on the species, so we have the simple algebraic equation
x + 3(−2) = −1
where x is the oxidation number of the nitrogen atom and −1 represents the charge on the species. Evaluating,
x + (−6) = −1
x = +5Thus, the oxidation number on the N atom in the nitrate ion is +5.
Test Yourself
Assign oxidation numbers to the atoms in H3PO4.
Answer
H = +1, O = −2, P = +5
All redox reactions occur with a simultaneous change in the oxidation numbers of some atoms. At least two elements must change their oxidation numbers. When an oxidation number of an atom is increased in the course of a redox reaction, that atom is being oxidized. When an oxidation number of an atom is decreased in the course of a redox reaction, that atom is being reduced. Oxidation and reduction are thus also defined in terms of increasing or decreasing oxidation numbers, respectively.
Example 14
Identify what is being oxidized and reduced in this redox equation.
2 Na + Cl2 → 2 NaCl
Solution
Consider the reactants. Because both reactants are the elemental forms of their atoms, the Na and Cl atoms as reactants have oxidation numbers of 0. In the ionic product, the sodium ions have an oxidation number of +1, while the chloride ions have an oxidation number of −1:
We note that the sodium is increasing its oxidation number from 0 to +1, so it is being oxidized; chlorine is decreasing its oxidation number from 0 to −1, so it is being reduced:
Because oxidation numbers are changing, this is a redox reaction. Note that the total number of electrons lost by the sodium (two, one lost from each atom) is gained by the chlorine atoms (two, one gained for each atom).
Test Yourself
Identify what is being oxidized and reduced in this redox equation.
C + O2 → CO2
Answer
C is being oxidized from 0 to +4; O is being reduced from 0 to −2.
In this introduction to oxidation-reduction reactions, we are using straightforward examples. However, oxidation reactions can become quite complex; the following equation represents a redox reaction:
To demonstrate that this is a redox reaction, the oxidation numbers of the species being oxidized and reduced are listed; can you determine what is being oxidized and what is being reduced? This is also an example of a net ionic reaction; spectator ions that do not change oxidation numbers are not displayed in the equation.
Key Takeaways
- Oxidation-reduction (redox) reactions involve the transfer of electrons from one atom to another.
- Oxidation numbers are used to keep track of electrons in atoms.
- There are rules for assigning oxidation numbers to atoms.
- Oxidation is an increase of oxidation number (a loss of electrons); reduction is a decrease in oxidation number (a gain of electrons).
Exercises
-
Is the reaction
2 K(s) + Br2(ℓ) → 2 KBr(s)
an oxidation-reduction reaction? Explain your answer.
-
Is the reaction
NaCl(aq) + AgNO3(aq) → NaNO3(aq) + AgCl(s)
an oxidation-reduction reaction? Explain your answer.
-
In the reaction
2 Ca(s) + O2(g) → 2 CaO
indicate what has lost electrons and what has gained electrons.
-
In the reaction
16 Fe(s) + 3 S8(s) → 8 Fe2S3(s)
indicate what has lost electrons and what has gained electrons.
-
In the reaction
2 Li(s) + O2(g) → Li2O2(s)
indicate what has been oxidized and what has been reduced.
-
In the reaction
2 Ni(s) + 3 I2(s) → 2 NiI3(s)
indicate what has been oxidized and what has been reduced.
-
What are two different definitions of oxidation?
-
What are two different definitions of reduction?
-
Assign oxidation numbers to each atom in each substance.
a) P4
b) SO2
c) SO22−
d) Ca(NO3)2
10. Assign oxidation numbers to each atom in each substance.
a) PF5
b) (NH4)2S
c) Hg
d) Li2O2 (lithium peroxide)
11. Assign oxidation numbers to each atom in each substance.
a) CO
b) CO2
c) NiCl2
d) NiCl3
12. Assign oxidation numbers to each atom in each substance.
a) NaH (sodium hydride)
b) NO2
c) NO2−
d) AgNO3
13. Assign oxidation numbers to each atom in each substance.
a) CH2O
b) NH3
c) Rb2SO4
d) Zn(C2H3O2)2
14. Assign oxidation numbers to each atom in each substance.
a) C6H6
b) B(OH)3
c) Li2S
d) Au
15. Identify what is being oxidized and reduced in this redox equation by assigning oxidation numbers to the atoms.
2 NO + Cl2 → 2 NOCl
16. Identify what is being oxidized and reduced in this redox equation by assigning oxidation numbers to the atoms.
Fe + SO3 → FeSO3
17. Identify what is being oxidized and reduced in this redox equation by assigning oxidation numbers to the atoms.
2 KrF2 + 2 H2O → 2 Kr + 4 HF + O2
18. Identify what is being oxidized and reduced in this redox equation by assigning oxidation numbers to the atoms.
SO3 + SCl2 → SOCl2 + SO2
19. Identify what is being oxidized and reduced in this redox equation by assigning oxidation numbers to the atoms.
2 K + MgCl2 → 2 KCl + Mg
20. Identify what is being oxidized and reduced in this redox equation by assigning oxidation numbers to the atoms.
C7H16 + 11 O2 → 7 CO2 + 8 H2O
Answers
1.
Yes; both K and Br are changing oxidation numbers.
3.
Ca has lost electrons, and O has gained electrons.
5.
Li has been oxidized, and O has been reduced.
7.
loss of electrons; increase in oxidation number
9.
a) P: 0
b) S: +4; O: −2
c) S: +2; O: −2
d) Ca: 2+; N: +5; O: −2
11.
a) C: +2; O: −2
b) C: +4; O: −2
c) Ni: +2; Cl: −1
d) Ni: +3; Cl: −1
13.
a) C: 0; H: +1; O: −2
b) N: −3; H: +1
c) Rb: +1; S: +6; O: −2
d) Zn: +2; C: 0; H: +1; O: −2
15.
N is being oxidized, and Cl is being reduced.
17.
O is being oxidized, and Kr is being reduced.
19.
K is being oxidized, and Mg is being reduced.